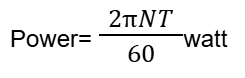FUNDAMENTALS OF THERMODYNAMICS
Other Chapters
FUNDAMENTALS OF THERMODYNAMICSLAWS AND PROPERTIES OF PERFECT GASESTHERMODYNAMIC PROCESS AND CYCLESFORMATION AND PROPERTIES OF STEAMSTEAM BOILERS AND PERFORMANCESTEAM AND GAS TURBINEINTERNAL COMBUSTION ENGINESAIR COMPRESSORS AND THEIR PERFORMANCEREFRIGERATION AND AIR CONDITIONING
- Q1: Define Thermodynamics.Ans: Thermodynamics is the branch of science that deals with heat, work, energy, and the relationship between them. It studies how energy is transferred and converted in different systems.
- Q2: Name the types of units.Ans: The types of units are:
- ➔ Basic Units
- ➔ Derived Units
- Q3: Define Basic Units.Ans: Basic units are the fundamental units which cannot be expressed in terms of other units. They form the base for all other units.
- Q4: Name some Basic Units.Ans: Some basic units are:
- ➔ Meter (m)
- ➔ Kilogram (kg)
- ➔ Second (s)
- ➔ Ampere (A)
- ➔ Kelvin (K)
- Q5: Define Derived Units.Ans: Derived units are the units which are obtained by combining two or more basic units according to physical laws.
- Q6: Name some Derived Units.Ans: Some derived units are:
- ➔ Newton (N)
- ➔ Joule (J)
- ➔ Watt (W)
- ➔ Pascal (Pa)
- Q7: Name the Systems of Units.Ans: The systems of units are:
- ➔ F.P.S System (Foot Pound Second System)
- ➔ M.K.S System (Meter Kilogram Second System)
- ➔ C.G.S System (Centimeter Gram Second System)
- ➔ S.I Units (System of International Units)
- Q8: Define Power.Ans: Power is the rate of doing work. It tells how fast work is done or energy is transferred.
Power = Work Done / Time - Q9: Write the unit of Power.Ans: The SI unit of power is Watt (W). One watt is equal to one joule of work done per second.
- Q10: Define System.Ans: A system is a definite portion of matter or space selected for study. Everything outside the system is called surroundings.
- Q11: Define Thermodynamic System.Ans: A thermodynamic system is a quantity of matter or a region in space chosen for thermodynamic study where energy transfer may occur.
- Q12: State the classification of System.Ans: The system is classified as:
- ➔ Closed System
- ➔ Open System
- Q13: Define Closed System.Ans: A closed system is a system in which mass does not cross the boundary, but energy transfer in the form of heat or work can occur.
- Q14: Define Open System.Ans: An open system is a system in which both mass and energy can cross the system boundary.
- Q15: What are the Extensive properties of a system?Ans: Extensive properties are the properties that depend on the quantity or size of the system. Examples include mass, volume, and total energy.
- Q16: What are the Intensive properties of a system?Ans: Intensive properties are the properties that do not depend on the size or quantity of the system. Examples include temperature, pressure, and density.
- Q17: Define Heat.Ans: Heat is a form of energy that is transferred from one body to another due to temperature difference.
- Q18: Define Heat Transfer.Ans: Heat transfer is the movement of heat energy from a higher temperature body to a lower temperature body.
- Q19: Name the methods of Heat Transfer.Ans: The methods of heat transfer are:
- ➔ Conduction
- ➔ Convection
- ➔ Radiation
- Q20: Define Conduction.Ans: Conduction is the method of heat transfer in which heat flows from one part of a body to another or from one body to another through direct contact, without movement of the particles.
- Q21: Define Convection.Ans: Convection is the method of heat transfer in which heat is transferred by the actual movement of fluid particles such as liquids or gases from one place to another.
- Q22: Define Radiation.Ans: Radiation is the method of heat transfer in which heat is transferred in the form of electromagnetic waves without the need of any material medium.
- Q23: Define Mass.Ans: Mass is the amount of matter present in a body. It remains constant irrespective of location.
- Q24: Define Weight.Ans: Weight is the force with which the earth attracts a body. It depends on gravitational acceleration.
- Q25: Define Gravitational Acceleration.Ans: Gravitational acceleration is the acceleration produced on a body due to the force of gravity. Its standard value on earth is 9.81 m/s².
- Q26: Define Force.Ans: Force is a push or pull that can change the state of rest or motion of a body. It is equal to mass multiplied by acceleration.
- Q27: Define Work.Ans: Work is done when a force moves a body through a certain distance in the direction of the force. The formula of Work is as under:
Work = Force x Distance - Q28: Define Pressure.Ans: Pressure is the force acting per unit area. It is equal to force divided by area.
- Q29: Define Normal Pressure.Ans: Normal pressure is the atmospheric pressure at sea level under normal conditions. It is equal to 1 atmosphere (1 atm) or 760 mm of mercury.
- Q30: Define Standard Pressure.Ans: Standard pressure is the reference pressure used for scientific calculations. It is equal to 1 atmosphere or 101.325 kPa.
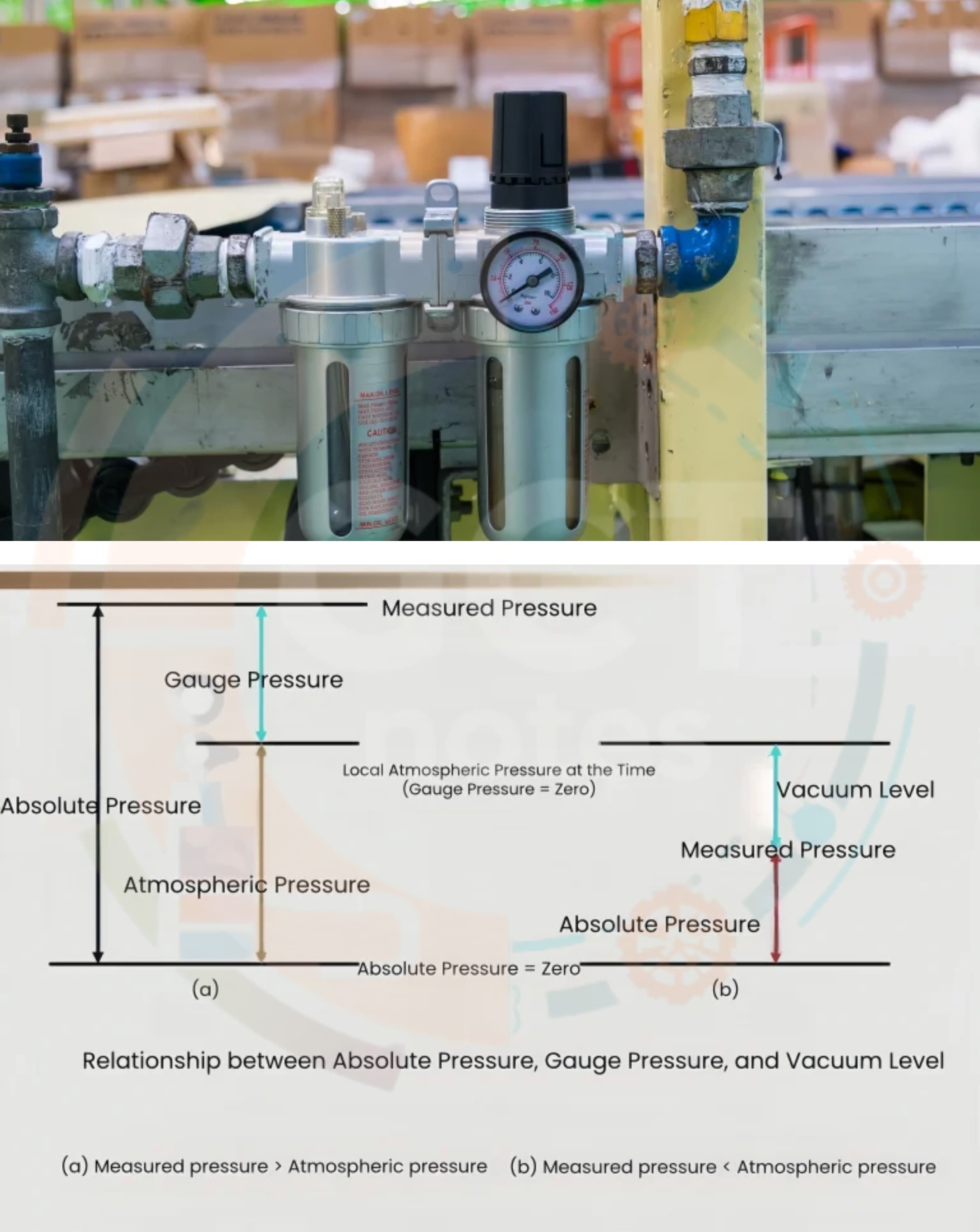
- Q31: Define Atmospheric Pressure.Ans: Atmospheric pressure is the pressure exerted by the weight of air present in the atmosphere on the surface of the earth.
- Q32: Define Vacuum Pressure.Ans: Vacuum pressure is the pressure below atmospheric pressure. It shows how much the pressure is less than the surrounding atmospheric pressure.
- Q33: Define Absolute Zero Pressure.Ans: Absolute zero pressure is the pressure measured from absolute vacuum. At this condition, there is no pressure because no molecules are present.
- Q34: Define Absolute Pressure.Ans: Absolute pressure is the total pressure measured from absolute zero pressure. It is equal to atmospheric pressure plus gauge pressure.
- Q35: Define Gauge Pressure.Ans: Gauge pressure is the pressure measured above atmospheric pressure. It does not include atmospheric pressure.
- Q36: Define Temperature.Ans: Temperature is the measure of the degree of hotness or coldness of a body. It indicates the level of heat energy present in a substance.
- Q37: Define Thermometer.Ans: A thermometer is an instrument used to measure the temperature of a body or substance.
- Q38: Define Gas Thermometer.Ans: A gas thermometer is a thermometer that measures temperature by observing the change in pressure or volume of a gas.
- Q39: Define Mercury Thermometer.Ans: A mercury thermometer is a thermometer that uses mercury as the working substance to measure temperature.
- Q40: Name the temperature scales.Ans: The temperature scales are:
- ➔ Celsius Scale
- ➔ Fahrenheit Scale
- ➔ Kelvin Scale
- Q41: Define Celsius or Centigrade Scale.Ans: Celsius or Centigrade scale is the temperature scale in which the freezing point of water is 0°C and the boiling point is 100°C at standard atmospheric pressure.
- Q42: Define Kelvin Scale.Ans: Kelvin scale is the absolute temperature scale in which 0 K represents absolute zero temperature. It is used in scientific calculations.
- Q43: Define Fahrenheit Scale.Ans: Fahrenheit scale is the temperature scale in which the freezing point of water is 32°F and the boiling point is 212°F at standard atmospheric pressure.
- Q44: Define Absolute Temperature.Ans: Absolute temperature is the temperature measured from absolute zero on the Kelvin scale.
- Q45: Define Normal Temperature.Ans: Normal temperature is the ordinary temperature of surroundings, generally taken as 0°C for reference in calculations.
- Q46: Define Standard Temperature.Ans: Standard temperature is the reference temperature used in scientific work, usually taken as 0°C or 273 K.
- Q47: Write the units of the followings.Ans:
Measuring Unit F.P.S M.K.S S.I Force Pound (lb) Kilogram-force (kgf) Newton (N) Distance Foot (ft) Meter (m) Meter (m) Time Second (s) Second (s) Second (s) Work Foot-pound (ft·lb) Kilogram-meter (kg·m) Joule (J) Velocity Foot per second (ft/s) Meter per second (m/s) Meter per second (m/s) Acceleration Foot per second² (ft/s²) Meter per second² (m/s²) Meter per second² (m/s²) Power Horsepower (HP) Kilogram-meter per second (kg·m/s) Watt (W) - Q48: Define Energy.Ans: Energy is the capacity of a system to do work. It can exist in various forms such as heat, mechanical, chemical, or electrical energy.
- Q49: Name the types of energy found in the system.Ans: The types of energy are:
- ➔ Stored Energy
- ➔ Transit Energy
- Q50: Define Stored Energy.Ans: Stored energy is the energy contained within a system at a given instant. It includes potential energy, kinetic energy, and internal energy.
- Q51: Define Transit Energy.Ans: Transit energy is the energy that crosses the boundary of a system in the form of work or heat during a process.
- Q52: Define Potential Energy.Ans: Potential energy is the energy possessed by a body due to its position or configuration relative to a reference point.
- Q53: Define Kinetic Energy.Ans: Kinetic energy is the energy possessed by a body due to its motion. It is proportional to the mass and square of velocity of the body.
- Q54: Define Internal Energy.Ans: Internal energy is the energy stored within a system due to the random motion and interactions of its molecules.
- Q55: State the laws of Thermodynamics.Ans: The laws of thermodynamics include: Zeroth Law, First Law, Second Law, and Third Law. These laws govern the behavior of energy, heat, and work in a system.
- Q56: Define Zeroth Law of Thermodynamics.Ans: Zeroth Law of Thermodynamics states that if two systems are in thermal equilibrium with a third system, then they are in thermal equilibrium with each other.
- Q57: Define First Law of Thermodynamics.Ans: The First Law of Thermodynamics states that energy can neither be created nor destroyed; it can only be transformed from one form to another. The change in internal energy equals heat added minus work done by the system.
- Q58: State the limitations of First Law of Thermodynamics.Ans: The First Law does not indicate the direction of energy transfer and cannot predict whether a process will occur spontaneously. It only quantifies energy changes.
- Q59: Define Second Law of Thermodynamics.Ans: The Second Law of Thermodynamics states that heat cannot spontaneously flow from a colder body to a hotter body, and all natural processes increase the entropy of the universe.
- Q60: What is Kelvin-Planck Statement?Ans: Kelvin-Planck Statement states that it is impossible to construct a heat engine that converts all the heat absorbed from a source into work without any other effect.
- Q61: What is Clausius Statement?Ans: Clausius Statement states that it is impossible for a process to transfer heat from a colder body to a hotter body without doing external work.
- Q62: State the Law of Conservation of Energy.Ans: The Law of Conservation of Energy states that the total energy of an isolated system remains constant; energy can change form but the total amount does not change.
- Q63: State the Law of Conservation of Matter.Ans: The Law of Conservation of Matter states that matter can neither be created nor destroyed in a chemical reaction; the total mass remains constant.
- Q64: What is the formula of Horse Power in M.K.S?Ans: Its formula is:
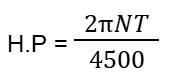
- Q65: What is the formula of Horse Power in F.P.S?Ans: Its formula is:
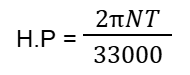
- Q66: What is the formula of Power in S.I?Ans: Its formula is: